BREAKING NEWS
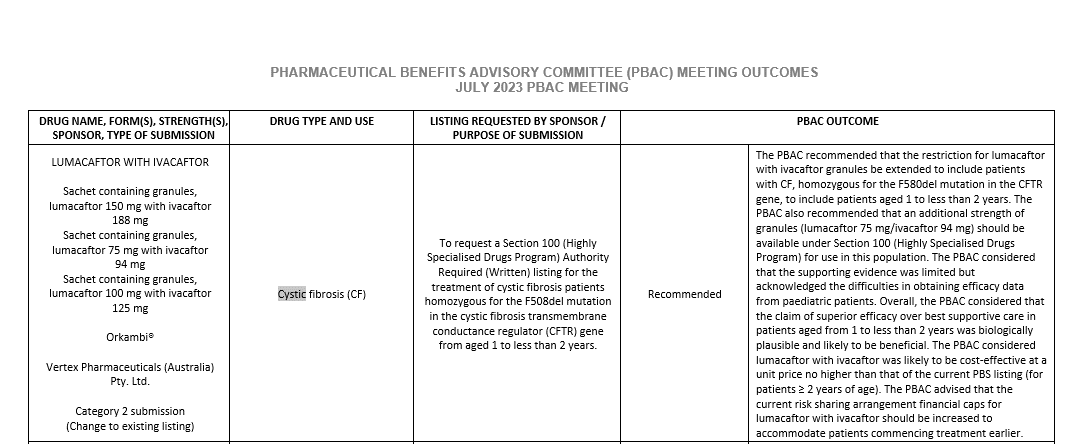
The CF modulator drug Orkambi (lumacaftor/ ivacaftor) has been recommended by the Pharmaceutical Benefits Advisory Committee (PBAC) for listing on the Pharmaceutical Benefits Scheme (PBS) for ages 1-2 in Australia.
This new therapy will benefit 35 children across Australia who have two copies of the F508del mutation.
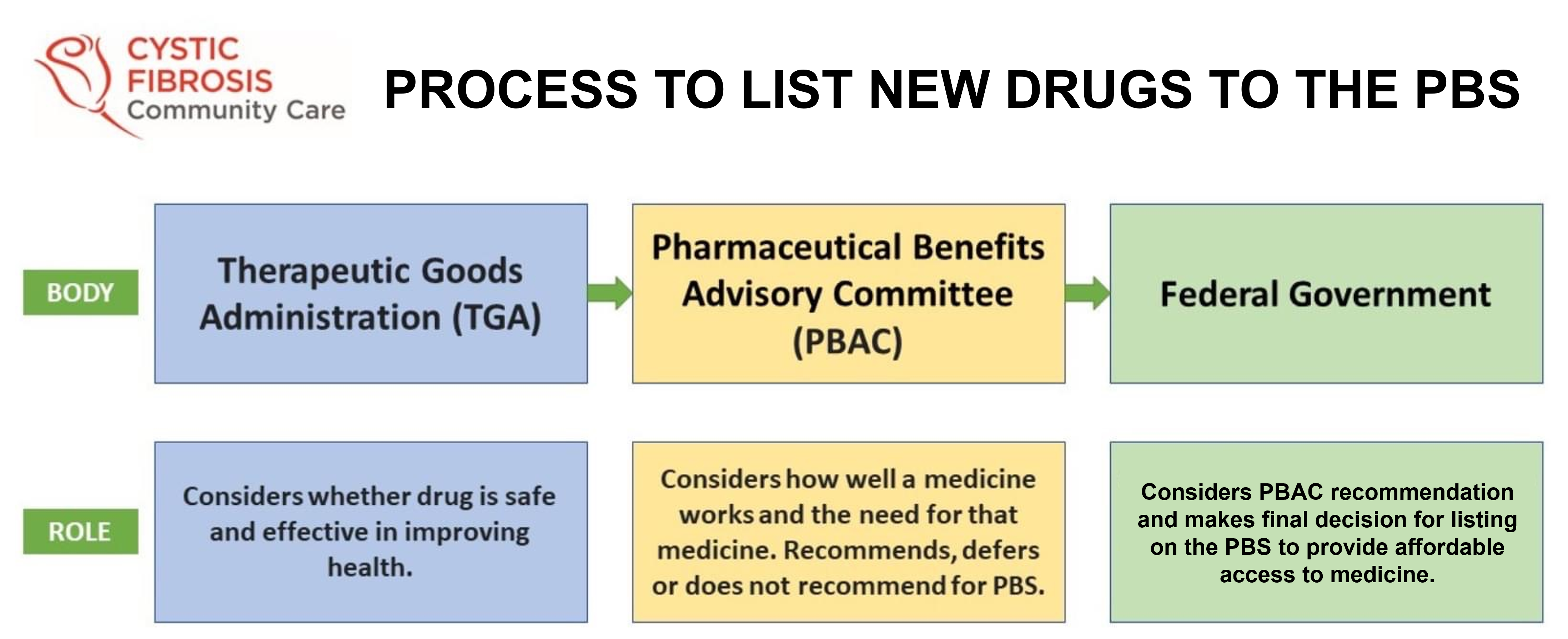
With this positive PBAC recommendation, the process moves to the Federal Government for final approval, and we wait with anticipation for this therapy to be made available as soon as possible.
Orkambi is a modulator therapy which has been available in Australia for those aged 2 years and older since 2019. In September last year, Orkambi was approved for use in the US for children aged 1-2 years, making it the only modulator therapy available for very young children with cystic fibrosis.
This therapy is available as a sachet, containing granules around the same size as flour particles, which can be mixed with a teaspoon of soft food or liquid so it can be consumed by young children.
About Orkambi
Cystic fibrosis is caused by changes in the cystic fibrosis transmembrane conductance regulator (CFTR) gene. This gene controls the production of CFTR protein. The CFTR protein creates channels on the cell surface to allow the movement of chloride in and out of the cell. When the CFTR protein is not working properly, the balance of chloride and fluids is affected, causing sticky mucus in organs such as the lungs and pancreas.
Orkambi is a modulator therapy developed by Vertex Pharmaceuticals, comprising of two active ingredients, lumacaftor and ivacaftor.
Ivacaftor binds to the defective CFTR protein at the cell surface and helps to open the channel so that chloride can flow in and out of the cells. This helps the balance of chloride and fluids at the surface of the cells and helps to thin mucus in the lungs and other organs. Lumacaftor helps the F508del-CFTR protein change to its correct shape, move to the surface of the cell, and stay there longer. Ivacaftor then helps to open the channel so that chloride can flow in and out of the cells.